Lesion Characteristics
Lesion Size (diameter): 12 mm
Lesion Location: Right upper lobe (RUL) lesion
Bronchus Sign: No
Visible on Fluoro: No
REBUS Verification: Yes, after Body Vision enabled navigation to the lesion
Case Information
Full Procedure Time: 45 minutes
ROSE: Sampling was performed and was positive for non-small cell lung cancer (NSCLC)
Final Pathology Report: Final pathology was adenocarcinoma of the lung
Patient Background
A 69 year-old asymptomatic, current smoker with COPD underwent a routine CT screening where a suspicious nodule was found in the right upper lobe (RUL). A PET scan of the 12 mm RUL nodule confirmed hyper-metabolic activity. A robotic-assisted navigation bronchoscopy procedure using the Auris Monarch platform in conjunction with Body Vision's AI-driven intraoperative imaging was scheduled to biopsy the nodule.
The Procedure
Planning
A pre-operative CT is uploaded onto the Body Vision AI-driven, intraoperative imaging system. The lesion is marked and contoured so that the Body Vision system has an approximation of lesion size, shape, and location prior to the procedure. The Body Vision system also derives the airways from the preoperative CT so for those looking to use the system as an end-to-end navigational bronchoscopy platform, a virtual bronchoscopy with preferred pathway to the lesion is provided. In this case, the Auris Monarch was used for navigation and so this functionality of the Body Vision system was not used.
Registration
Once the patient is on the table, Body Vision registration consists of two C-arm spins: one around the main carina and a second iso-centered on the lesion. This produces an initial C-Arm Based Computed Tomography (CABT) scan to confirm actual lesion and lesion location prior to navigation. Once this is complete, registration of the navigation platform of choice, in this case, the Auris Monarch, is performed.
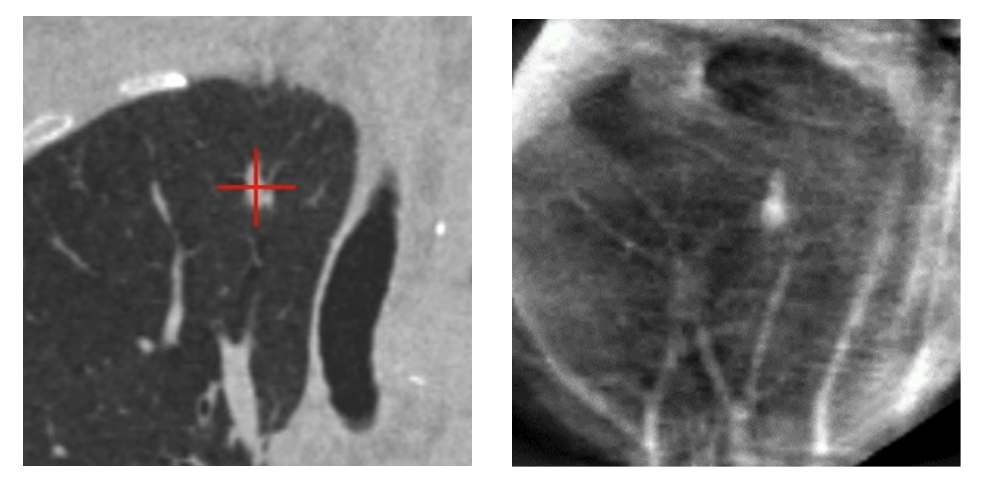
Pre-operative CT in coronal view showing 12mm RUL lesion (left). Body Vision 3D tomographic scan (right) during registration confirming actual size, shape, and location of lesion prior to navigation.
Navigation
The Auris Monarch robotic bronchoscopy platform was navigated to the virtual target as defined by the robotic platform’s electromagnetic navigation (EMN) technology. A radial endobronchial ultrasound (r-EBUS) probe was advanced down the working channel of the Monarch bronchoscope to validate accuracy of the virtual target location. The r-EBUS did not return a signal indicative of a lesion at the virtual target location.
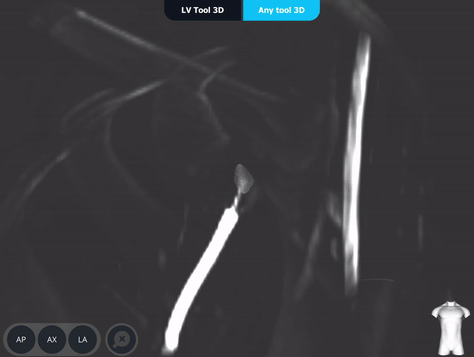
A GE OEC 9900 Elite C-arm was brought into position over the patient and a CABT scan with the Body Vision system was performed to determine true lesion location. The intraoperative CT image overlaid onto the Body Vision-generated augmented fluoroscopy enabled repositioning of the robotic bronchoscope. Because Body Vision’s imaging is not based on preoperative CT scans and is captured in real-time intraoperatively, no CT-to-body divergence exists.
Using Body Vision’s fluoroscopy augmented with lesion location as visualized on the most recent C-arm spin for real-time, intraoperative guidance, the Auris Monarch robotic bronchoscope was repositioned to the actual lesion location.
r-EBUS Confirmation
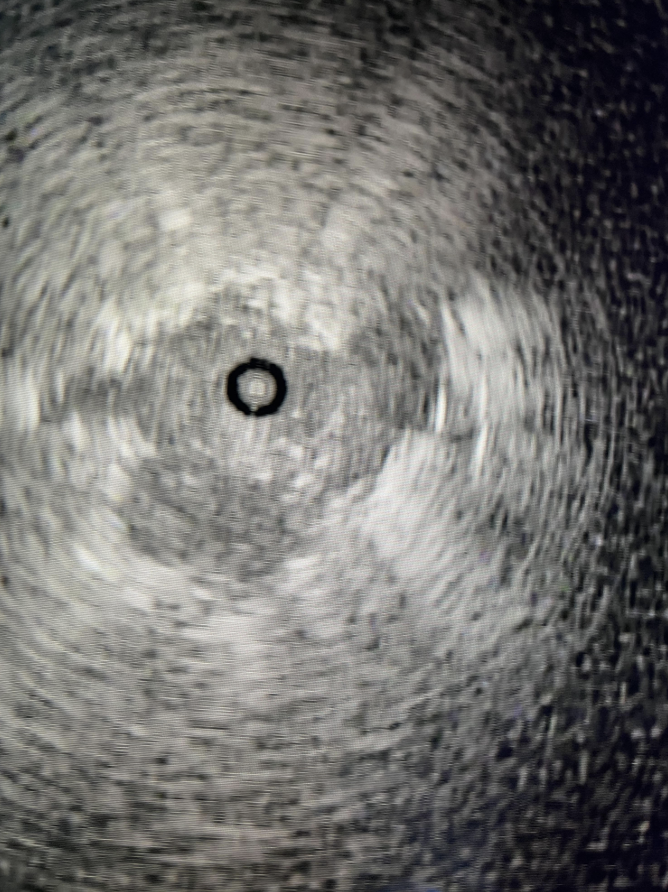
R-EBUS probe confirming tool-in-lesion.
The r-EBUS probe was re-inserted down the working channel of the Monarch robotic bronchoscope and extended into the lesion location as shown by Body Vision. At the new bronchoscope location, the r-EBUS probe returned a concentric echo-density, supporting that the lesion location as defined by Body Vision was correct.
Tool-in-Lesion Confirmation
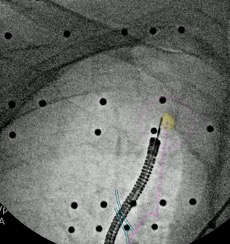
The r-EBUS probe was retracted from the Monarch working channel and replaced with biopsy forceps which were extended into the pulmonary lesion as visualized with Body Vision and verified by r-EBUS. Another C-arm spin was performed with the biopsy forceps extended in order to provide visual confirmation of tool-in-lesion in multiple 3D planes before biopsy.
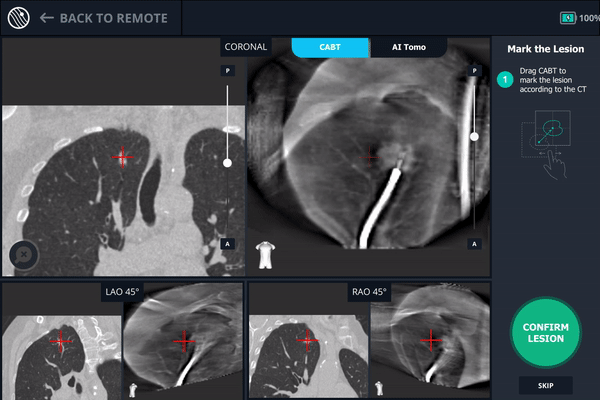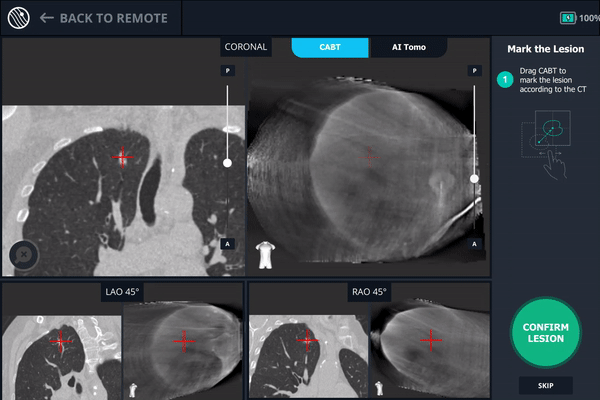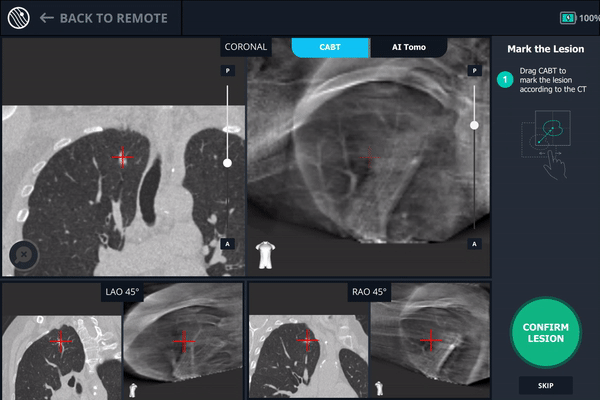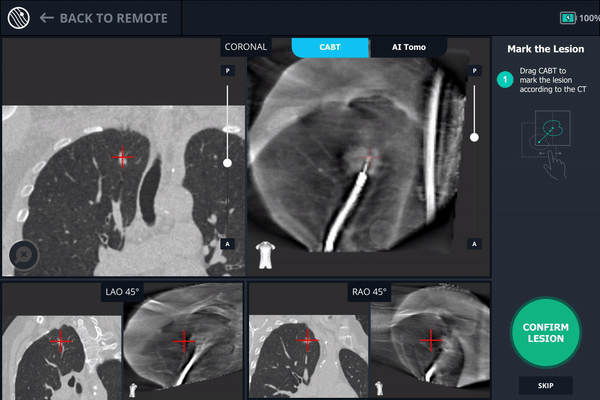
Body Vision’s AI-driven imaging enabled a C-arm to produce real-time intraoperative scans that visualize the lesion, bronchoscope, and biopsy tools for tool-in-lesion confirmation prior to biopsy.
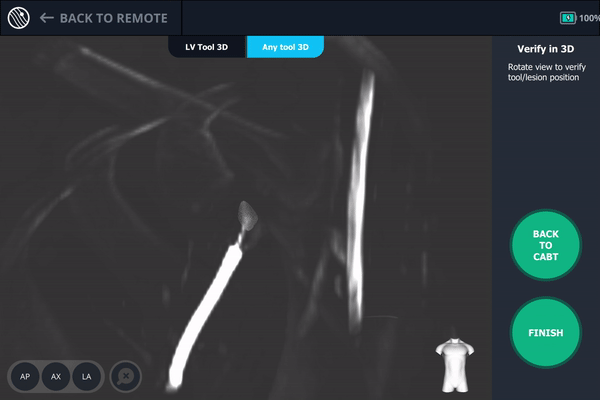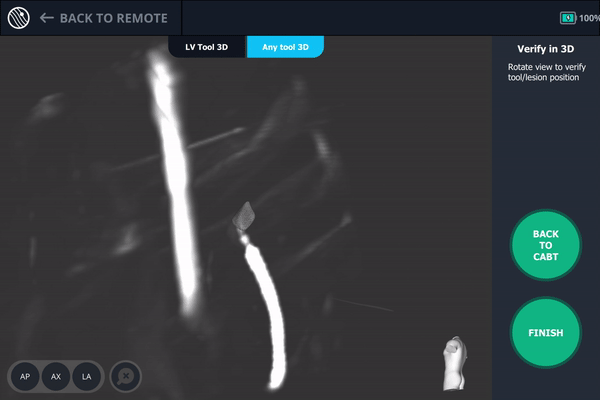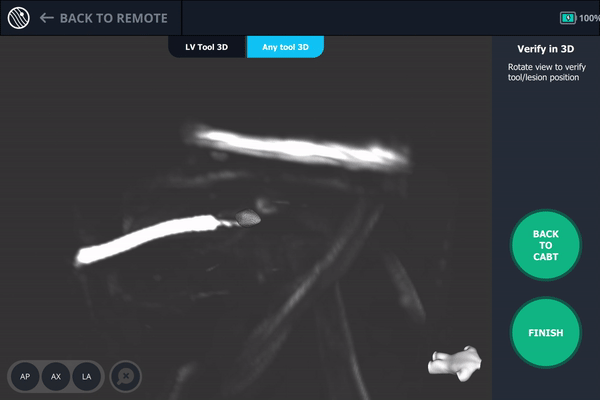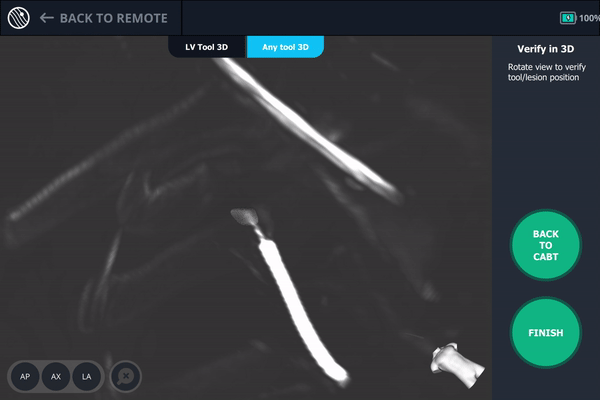
Body Vision's 3D View provided an additional means for visually confirming tool-in-lesion.
Biopsy
After tool-in-lesion confirmation, biopsy commenced under real-time image guidance using Body Vision's augmented fluoroscopy to ensure that tissue sampling occurred from within the lesion. Rapid On-Site Evaluation (ROSE) assessed that samples collected were adequate and consistent with non-small cell lung cancer. This was later confirmed by cytology on the final pathology report which stated adenocarcinoma of the lung.
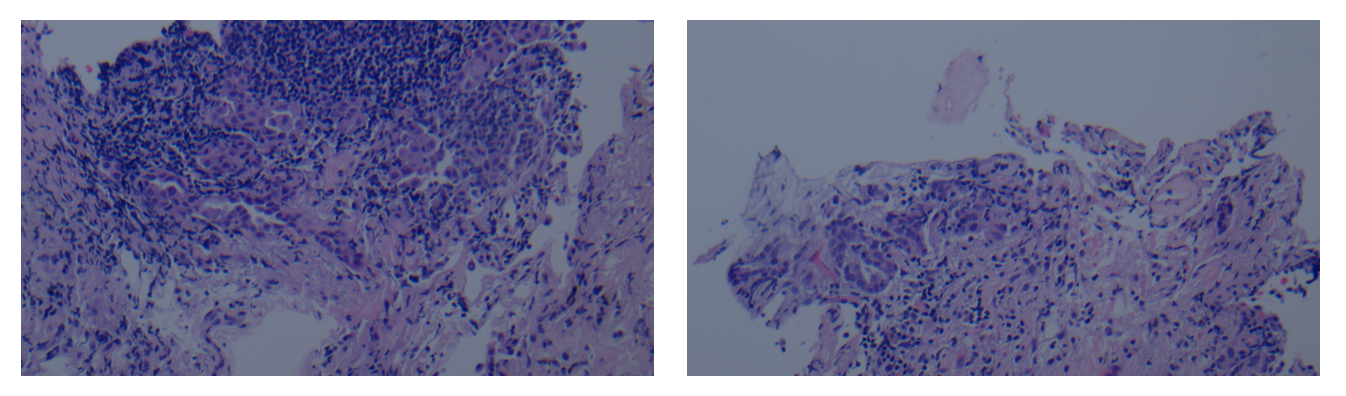
Final pathology report indicating adenocarcinoma of the lung.
A fiducial marker was placed at this time. Due to poor lung function (FEV1 less than 800 cc's), this patient is not an operative candidate. EBUS performed at the end of bronchoscopy presented no evidence of hilar or mediastinal lymph node involvement. This patient is an excellent candidate for Cyberknife.
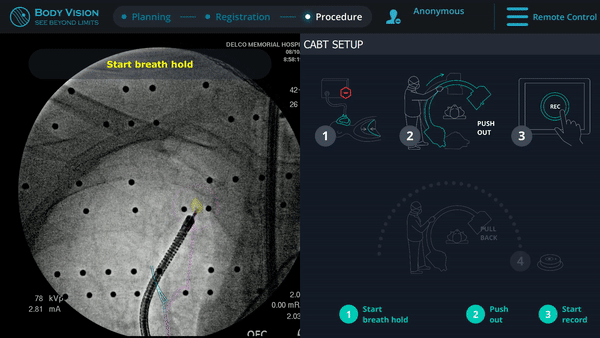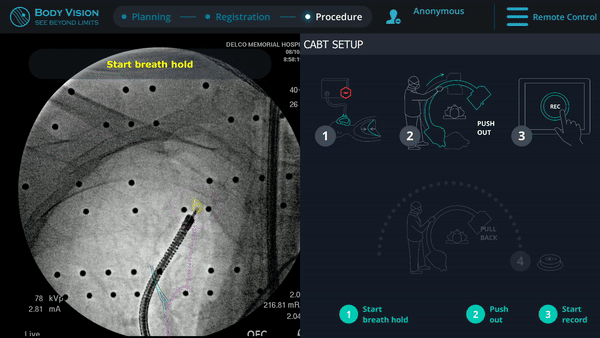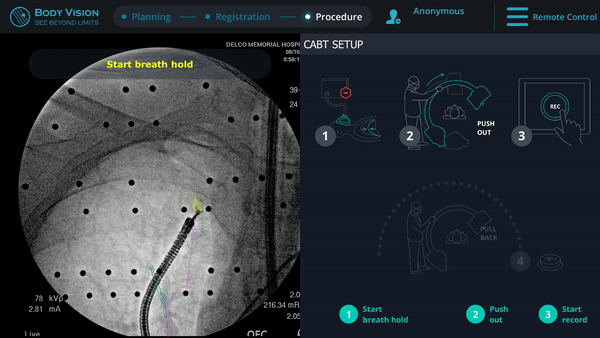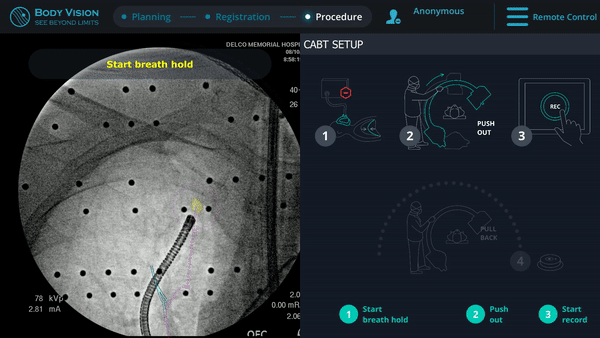
Body Vision's augmented fluoroscopy showing real-time biopsy via forceps.
Conclusion
This case illustrates how Body Vision’s AI-driven, intraoperative imaging can be seamlessly incorporated into the generally-accepted diagnostic bronchoscopy workflow. While robotic assisted bronchoscopy lends added stability and articulation to navigation bronchoscopy, the lack of real-time, intraoperative 3D imaging prevents any potential correction should, as in this case, the virtual target provided by the Monarch’s EMN navigation system prove to be incorrect due to CT-to-body divergence or other factors.
Rather than resorting to systematic blind sampling of the lung with r-EBUS to find the lesion, Body Vision enabled visualization of the lesion and lesion location so that the bronchoscope could be repositioned to the actual lesion location. Once the bronchoscope was repositioned, Body Vision’s intraoperative imaging enabled visual tool-in-lesion confirmation to maximize likelihood that tissue samples obtained were from within the lesion. Real-time, intraoperative imaging transformed this case from one in which reliable sampling from within the pulmonary lesion seemed unlikely to one in which a definitive diagnosis of adenocarcinoma of the lung was obtained for this lung patient.
About Dr. Whitlark

Joseph D. Whitlark, MD
Robotic Thoracic Surgeon
Crozer Health