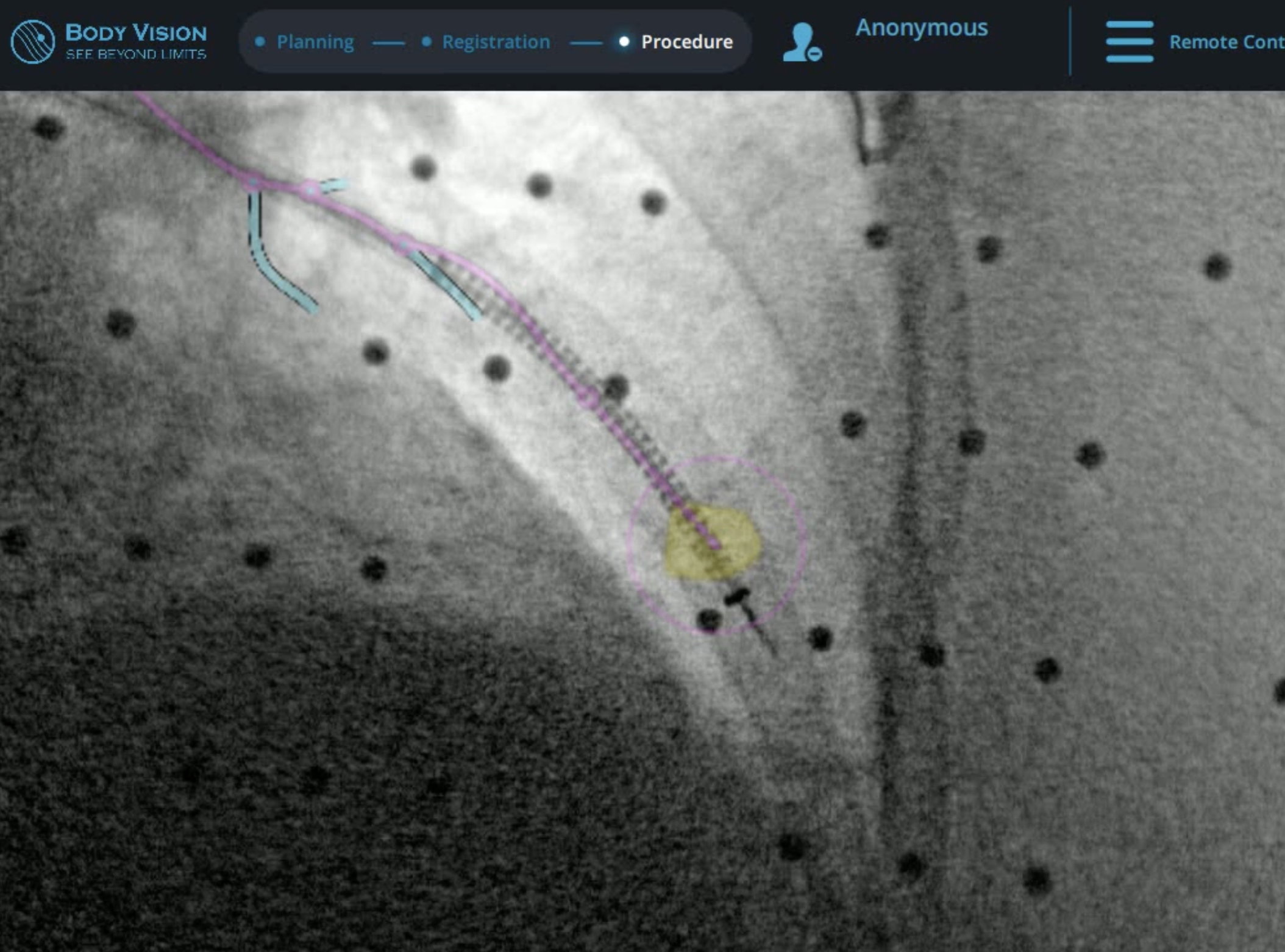
Overcoming CT-to-Body Divergence
CT-To-Body Divergence Explained
CT-to-body divergence is the result of the difference between the anatomy as it is represented in static preprocedural CT scans and the anatomy in the dynamic, breathing lung during the bronchoscopic procedure. This divergence is problematic when attempting to obtain a biopsy sample from within a suspicious lesion because static imagery gained from preoperative CT scans is not necessarily representative of where the actual lesion is during the bronchoscopy procedure due to differences in patient position, atelectasis, innate dynamics of the lung anatomy, and other factors.
This can result in a several-centimeter difference in location between where the lesion appears in the preoperative CT and where the lesion actually is located during the procedure. This is not a trivial amount of error when one considers that the average lung nodule size that bronchoscopists attempt to biopsy from is 2cm in size or less. Without taking CT-to-body divergence into account, accuracy is sacrificed, and the ability to successfully diagnose the patient is at risk. Addressing CT-to-body divergence is necessary for reliable diagnostic outcomes.
What Contributes to CT-To-Body Divergence?
Preprocedural Factors
Lung Volume Variance
Structure mapping from a preprocedural computed tomography (CT) scan varies significantly based on the current patient’s lung volume. Ideally, the patient’s imaging would be done while their lungs are fully inflated after an inhale.1 The procedure would then be completed while the patient’s lungs were again at full self-inflated volume.
However, this poses an issue due to the nature of the respiratory cycle. As a patient naturally breathes in and out with different lung volumes throughout the respiratory cycle, the location of internal pulmonary structures will shift with this movement.2 This means even the most accurate static preprocedural CT will be somewhat different than what will be seen under intraoperative imaging during the procedure.3
Computed Tomography Scan Scheduling
Preprocedural CT scans are generally conducted anywhere from days to months before a scheduled procedure. In two separate studies, 7-10% of cases were shown to exhibit decreases in the size of lung lesions as noted on initial CT imaging.4, 5 In a similar study of the Veran system, 6.9% of nodules decreased in size or disappeared altogether before the bronchoscopy procedure took place, over an average of 53 days.6 This means that there is a good chance that lungs on the day of procedure will look different than they did when the preprocedural CT scan was captured.
Patient Position
Anatomical variances due to patient position can also contribute to CT-to-body divergence. Patients positioned with their arms overhead, versus by their side, will change the location of thoracic and lung anatomy as well as lesion location. The bed position, using a pillow, and an arched spine in the patient while lying down can also affect the imaging process.
Magnetic Interference
In navigation systems that use electromagnetics, such as electromagnetic navigation (EMN) bronchoscopy, magnetic materials can cause errors if located within range of the magnetic field. Anesthesia equipment, intravenous poles, fluoroscopy units, beds, and any other ferromagnetic object should be closely managed to ensure there is no interference.
Procedural Factors
Atelectasis
Atelectasis can cause lung mapping errors and decrease the visibility of lesions on imaging. Studies show that atelectasis occurs in 87-100% of healthy adult patients within minutes of general anesthesia induction.7 Atelectasis is more common in obese individuals and those with scoliosis. Atelectasis can also be caused due to prolonged time to intubation, suboptimal ventilation procedure, and high fractions of inspired oxygen. One study shows some level of atelectasis was identified intraoperatively by cone-beam CT (CBCT) in 40% of total cases and that atelectasis completely obscured the lesion in 20% of cases.8 The anesthesia method used, as well as patient anatomy and weight, are likely contributing causes..
Another cause of atelectasis is bronchoscopy tools and airway manipulation during procedures. Often, the bronchoscope can be wedged in the airway, creating an obstruction leading to obstructive atelectasis and possible airway distortion.
Applying suction can exacerbate this issue, along with endoscopic tools used during lung nodule sampling that often displace or distort patient anatomy. The flexibility of catheters and other instruments will contribute to the variation here.
Often, advanced bronchoscopists and interventional pulmonologists stage the mediastinum before sampling the lung nodule, which extends the length of time the patient is supine and under anesthesia. This can sometimes cause de-recruitment of the peripheral alveoli and additional atelectasis, especially near the diaphragm. Atelectasis and anatomical changes can continue during the procedure itself, causing a dynamically shifting structure that varies greatly from the original imaging. These effects are most common in the lower lobes but can affect the upper lobes as well.9
Flexible catheters and instruments may help decrease airway deflection. It is now recommended that navigation bronchoscopy be conducted before mediastinal staging to reduce CT-to-body divergence during the procedure, as long as there is no suspicion of mediastinal metastasis.
Chest Muscle Variation
In order to reduce CT-to-body variance, paralytics may be considered. Neuromuscular blocking agents affected the effector muscles, especially those in the upper airway. 10 The nondependent part of the lung has been shown to be most displaced after neuromuscular bocks and positive pressure ventilation.10 Paralytic agents don’t affect the diaphragm muscles to a large degree, and they have a much greater effect on abdominal muscles. This is why ventilation strategies are essential to minimize lung motion in addition to the use of any paralytics.11 Relaxation of the expiratory abdominal muscles and intercostal muscles can change the conformation of the chest wall and may also contribute to CT-to-body divergence.10 This effect should be considered when using paralytics and inhaled anesthetics.
Practical Implications
Body Vision’s intraoperative imaging eliminates CT-to-body divergence by enabling physicians to see that they are sampling from within the lesion. With access to real-time, intraoperative imaging, Body Vision is enabling physicians to more confidently diagnose lung nodules.. This leads to earlier diagnosis and a higher likelihood of survival for lung cancer patients.
Traditionally, lung cancer was detected incidentally on chest X-rays.. In order to confirm malignancy, a biopsy must be performed. One common means of obtaining a biopsy is the transthoracic needle aspiration (TTNA) procedure, in which an interventional radiologist enters the lung through the chest wall with a needle to obtain a sample. TTNA, while less invasive than surgery, still has a relatively high complication rate and limitations as to which nodules can be biopsied in this fashion.
Alternatively, diagnostic bronchoscopy uses a minimally invasive procedure where a flexible tube with a small camera located at the end called a bronchoscope is navigated through the mouth, down the trachea and into the lungs in order to get a biopsy. While this procedure is significantly less invasive, without real-time, intraoperative imaging, the diagnostic rate is typically lower than TTNA.
In addition to the risk associated with these diagnostic procedures and biopsy methods, extended hospital stays, extra recovery time, and additional costs are possible. Body Vision’s AI-driven, intraoperative imaging offers a revolutionary opportunity to diagnose cancerous peripheral pulmonary lesions via a minimally invasive procedure.
How It Works
Body Vision’s intraoperative imaging technology utilizes a proprietary AI imaging algorithm to transform preexisting imaging equipment in the procedure room into a real-time, intraoperative imaging system. This system enables the physician to see the actual lung lesion and lesion location in a way they previously were unable to during the procedure. Body Vision’s intraoperative imaging solution integrates with any conventional C-arm to provide high-quality, CT-like imaging with just a simple C-arm spin. Because scans are captured during the procedure, any CT-to-body divergence is eliminated and bronchoscopists can be confident that they are sampling from within the lesion. Body Vision’s imaging is clinically proven to maximize diagnostic yield, up to 91%.12
Sources
- Furukawa, B. S., Pastis, N. J., Tanner, N. T., Chen, A., & Silvestri, G. A. (2018). Comparing pulmonary nodule location during electromagnetic bronchoscopy with predicted location on the basis of two virtual airway maps at different phases of respiration. Chest, 153(1), 181–186. https://doi.org/10.1016/j.chest.2017.06.004
- Kolar P, Neuwirth J, Sanda J, et al. (2009). Analysis of diaphragm movement during tidal breathing and during its activation while breath holding using MRI synchronized with spirometry. Physiol Res 2009;58:383-92.
- Eberhardt R, Anantham D, Ernst A, et al. (2007). Multimodality bronchoscopic diagnosis of peripheral lung lesions: a randomized controlled trial. Am J Respir Crit Care Med 2007;176:36-41. 10.1164/rccm.200612-1866OC
- Semaan RW, Lee HJ, Feller-Kopman D, et al. (2016). Same-Day Computed Tomographic Chest Imaging for Pulmonary Nodule Targeting with Electromagnetic Navigation Bronchoscopy May Decrease Unnecessary Procedures. Ann Am Thorac Soc 2016;13:2223-8. 10.1513/AnnalsATS.201607-522BC
- Zhao YR, Heuvelmans MA, Dorrius MD, et al. (2014). Features of resolving and nonresolving indeterminate pulmonary nodules at follow-up CT: the NELSON study. Radiology2014;270:872-9. 10.1148/radiol.13130332
- Semaan RW, Lee HJ, Feller-Kopman D, et al. (2016). Same-Day Computed Tomographic Chest Imaging for Pulmonary Nodule Targeting with Electromagnetic Navigation Bronchoscopy May Decrease Unnecessary Procedures. Ann Am Thorac Soc 2016;13:2223-8. 10.1513/AnnalsATS.201607-522BC
- Lundquist H, Hedenstierna G, Strandberg A, et al. (1995). CT-assessment of dependent lung densities in man during general anaesthesia. Acta Radiol1995;36:626-32. 10.1177/028418519503600464
- Casal RF, Sarkiss M, Jones AK, et al. (2018). Cone beam computed tomography-guided thin/ultrathin bronchoscopy for diagnosis of peripheral lung nodules: a prospective pilot study. J Thorac Dis2018;10:6950-9. 10.21037/jtd.2018.11.21
- Chen A, Pastis N, Furukawa B, et al. (2015). The effect of respiratory motion on pulmonary nodule location during electromagnetic navigation bronchoscopy. Chest 2015;147:1275-81. 10.1378/chest.14-1425
- Froese AB, Bryan AC. (1974). Effects of anesthesia and paralysis on diaphragmatic mechanics in man.Anesthesiology 1974;41:242-55. 10.1097/00000542-197409000-00006
- Kleinman BS, Frey K, VanDrunen M, et al. (2002). Motion of the diaphragm in patients with chronic obstructive pulmonary disease while spontaneously breathing versus during positive pressure breathing after anesthesia and neuromuscular blockade. Anesthesiology2002;97:298-305. 10.1097/00000542-200208000-00003
- Hedstrom G and Wagh A. Combining Real-Time 3D Imaging and Augmented Fluoroscopy with Robotic Bronchoscopy for Diagnosis of Peripheral Lung Nodules. Chest, Volume 162, Issue 4, Supplement, 2022, Page A2082