Lesion Characteristics
Lesion Size (diameter): 10 mm
Lesion Location: Right upper lobe (RUL) lesion
Bronchus Sign: No
Visible on Fluoro: Yes
REBUS Verification: Yes
Case Information
Full Procedure Time: 25 minutes
ROSE: Sampling was performed and was malignant
Final Pathology Report: Squamous cell lung carcinoma
Patient Background
A 69 year-old female with severe COPD, a lifetime smoking history, scoliosis, rheumatoid arthritis, hypertension and thrombocytosis had a screening CT and subsequent PET that revealed a 10 mm hyper-metabolic nodule in the right upper lobe.
The Procedure
Planning
During planning, a preoperative CT is loaded onto the Body Vision system. The lesion is marked and contoured so that Body Vision’s AI-driven intraoperative imaging system has an approximation of the lesion size, shape and location prior to beginning the procedure. The Body Vision system also derives the airways from the preoperative CT and for those who are looking for a complete navigation bronchoscopy system, a virtual bronchoscopy highlighting the preferred pathway to the lesion is provided. In this case, an Ethicon MONARCH(™) robotic bronchoscopy platform was used to navigate out to the lesion.
Registration
Once the patient is on the table, Body Vision registration consists of two C-arm spins – one iso-centered around the main carina and another iso-centered around the lesion. These spins produce an initial C-Arm Based Computed Tomography (CABT) scan to confirm actual lesion and lesion location on the day of procedure prior to navigation.
Marking the main carina on Body Vision’s CABT during registration.
Marking the lesion on Body Vision’s CABT during registration.
Once this is complete, registration of the Body Vision system is finished and registration of the MONARCH robot can now be performed.
Navigation
The MONARCH robotic bronchoscopy platform was used to navigate out to the virtual target. Once at the target location defined by MONARCH, a radial endobronchial ultrasound (r-EBUS) probe was inserted down the working channel of the MONARCH. The r-EBUS returned a signal consistent with the nodule so no additional tool-in-lesion spin was performed.
Biopsy
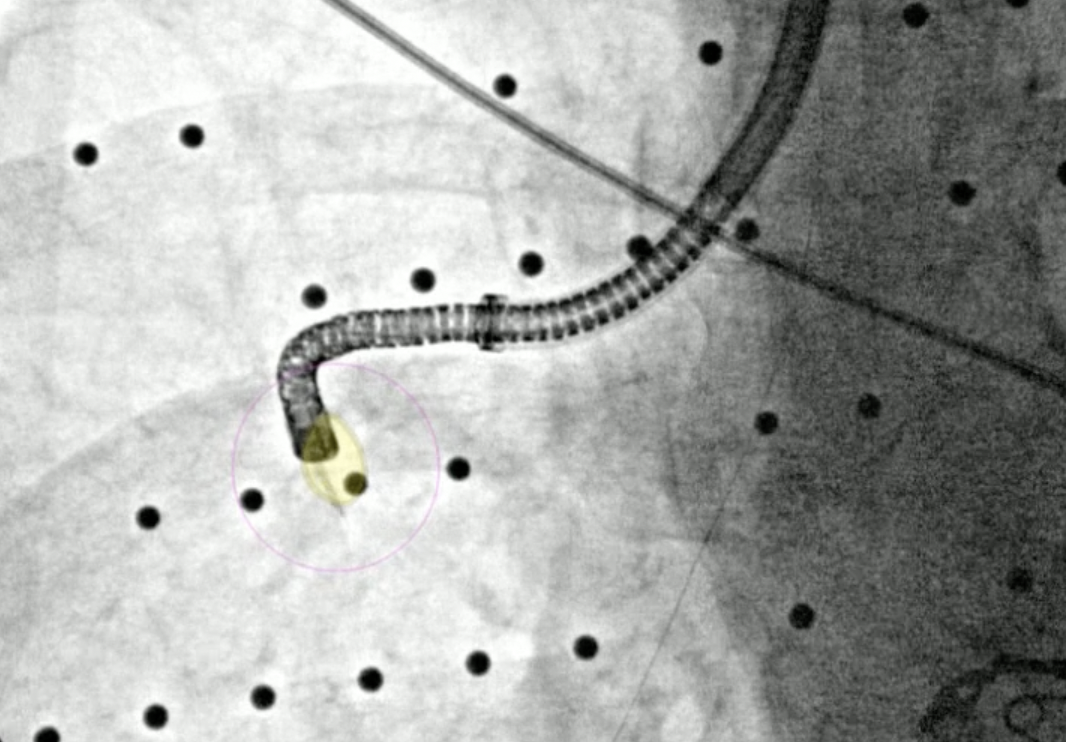
After the r-EBUS probe returned a positive signal, biopsy samples via needle and forceps were acquired under Body Vision’s real-time, augmented fluoroscopy. A fiducial marker was also placed using guidance from Body Vision’s augmented fluoroscopy. Rapid On-Site Evaluation (ROSE) confirmed that the tissue samples were malignant. The final pathology report stated squamous cell lung carcinoma.
Needle biopsy performed under Body Vision’s augmented fluoroscopy.
Another tissue sample acquired via forceps under Body Vision’s augmented fluoroscopy.
A fiducial marker being placed under Body Vision’s augmented fluoroscopy.
Final Pathology
Following the final pathology report confirming Squamous Cell Lung Carcinoma, this patient was sent for Stereotactic Body Radiation Therapy (SBRT).
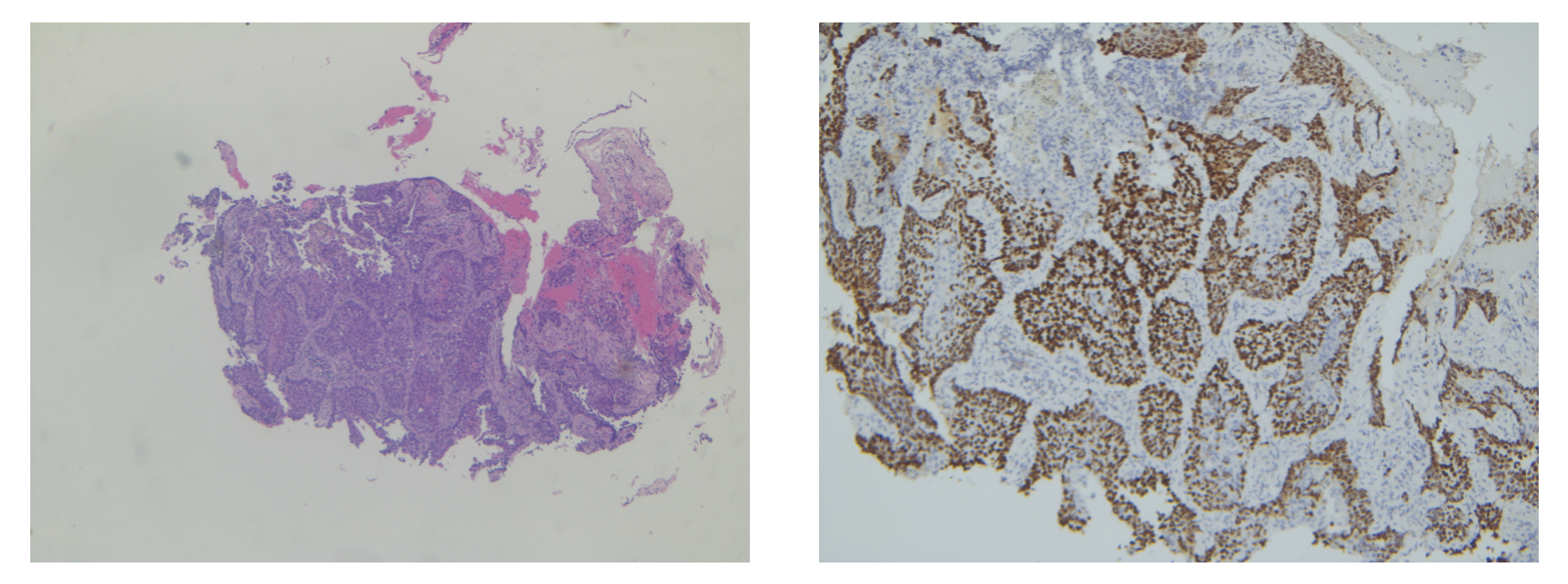
Final pathology report indicating squamous cell lung carcinoma.
Conclusion
This case demonstrates the ability of Body Vision’s AI-driven, intraoperative imaging system to work seamlessly with the Ethicon MONARCH robotic bronchoscope in a cohesive diagnostic bronchoscopy workflow while adding the missing component of real-time, intraoperative imaging necessary to confirm that tissue samples are taken from within the lesion during biopsy. In this 25-minute case, Body Vision enabled the biopsy, definitive diagnosis of and fiducial marker placement within this small, centimeter lesion.
About Dr. Whitlark
Joseph D. Whitlark, MD
Robotic Thoracic Surgeon
Crozer Health